Massachusetts Institute of Technology Massachusetts Eye and Ear Infirmary, MIT-Lincoln Laboratories, Southern College of Optometry
Purpose. Quantification of the electrical parameters needed to initiate action potentials from extracellular stimulation of rabbit retinal ganglion cells, to estimate the power needed for an epiretinal prosthesis.
Methods. A retinal strip with attached pigment was isolated and superfused. A recording electrode was used to identify action potentials of individual axons; receptive field centers driving the axons were located in the peripheral retina. Monopolar electrodes were lowered onto the surface of the retina within the receptive field center, and the threshold needed to generate an action potential was determined using monophasic, cathodal stimulation for 20, 50, 100, 200, and 500ps, An X-Y-Z grid was made of the region surrounding the receptive field center for 13 retinal ganglion cells.
Results. Thresholds increased 6 fold when the stimulation duration was decreased to 20,us from 500,us. Based partly upon this result, stimulation durations of 100ps were used for grid mapping. Thresholds at the retinal surface in the center of the receptive field ranged from B- 24,uA (mean: 14. 1 t6.5) for Pt/Ir electrodes and 1 1 -53,UA mean:27.6 tl 7.3) for tungsten electrodes. Along X and Y axes of 6 cells, thresholds increased 1.7 fold when the electrode was positioned 50A from receptive field center. Thresholds increased 2.5 fold when the electrode was positioned 50,u above the retina, and 4.5 fold at 1 00,u above the retina.
Conclusions. Epiretinal stimulation of retinal ganglion cells in the rabbit can be achieved with relatively low current, within a range that can be supplied by an implantable photodiode array.

Figure 1 We have measured the thresholds for electrical stimulation of rabbit retinal ganglion cells to aid in the design of a silicon retinal prosthesis. The specific goals were to:
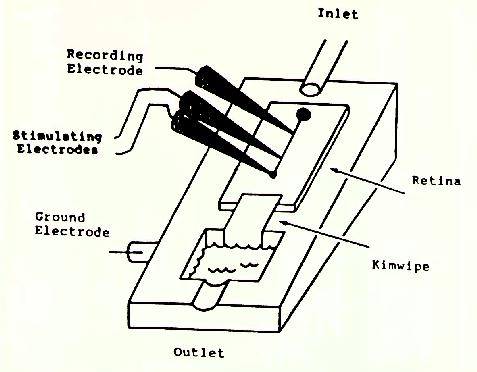
Figure 2

Figure 3
Experiments like those shown in Figure 5 were performed on 37 ganglion cells and on the axons of 30 of these cells. The minimum threshold obtained as the electrode was moved across the retina is plotted here in histogram form. The mean axonal threshold was 15.9 microamps and the mean cell body threshold was 8.3 microamps. Unfortunately the threshold populations overlap substantially, making it impossible to find a current intensity that would selectively stimulate ganglion cells alone.
Stimulation threshold for ganglion cells increases with vertical (z-axis) displacement from cell body and horizontal (x and y axes) displacement. The plot in Figure 6 shows ratio of measured threshold at various positions to the minimum threshold observed in experiments like those shown at the top of this panel, These preliminary data for horizontal displacements of the electrode from 6 ganglion cells and vertical displacements from 7 cells show that thresholds rise only moderately for ou micron displacements in any direction, but go up steeply for vertical displacements of 100 microns. Data were obtained using 100 microsecond cathodal pulses applied through 10 micron platinum-iridium electrodes.
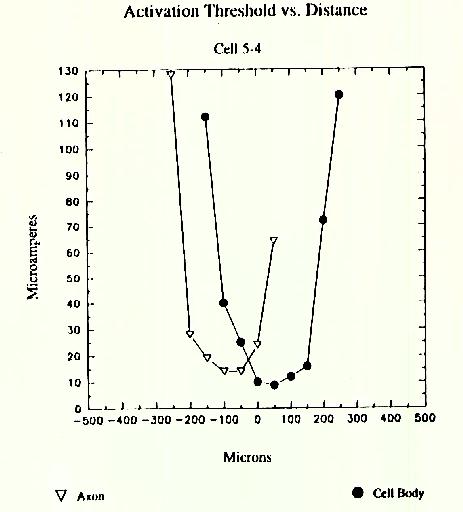
Figure 4
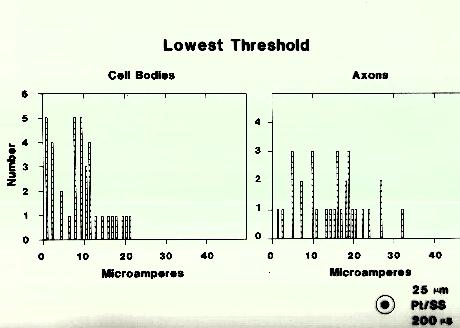
Figure 5
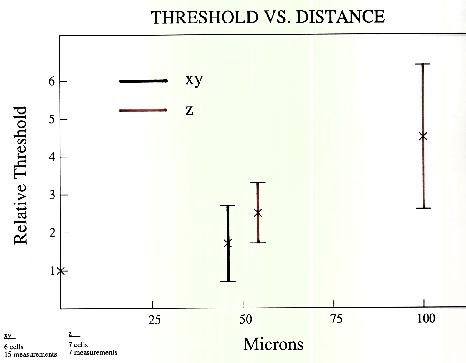
Figure 6 In the measurements recorded in Figure 7 and Figure 8 (following page) the electrode was positioned over each point in a 250 micron x 250 micron grid with 25 micron spacing. It was lowered to the point of lowest threshold for anodal stimulation, and the values are recorded in the left colored plot above. The cathodal thresholds were recorded in the same locations and appear in the right colored plot above. The lowest thresholds were observed at the center point of each plot, where they are given in each figure in microamps. The highest values are also given in microamps at the point where they were observed. The plots are quite assymetric, with lower thresholds observed along a line from the center to the upper right corner. Data were obtained using 100 microsecond pulses applied through a 5 micron platinum-iridium electrode.
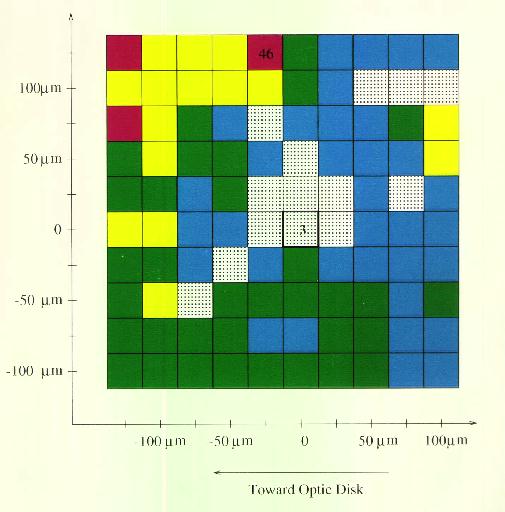
Figure 7 Anodal Thresholds
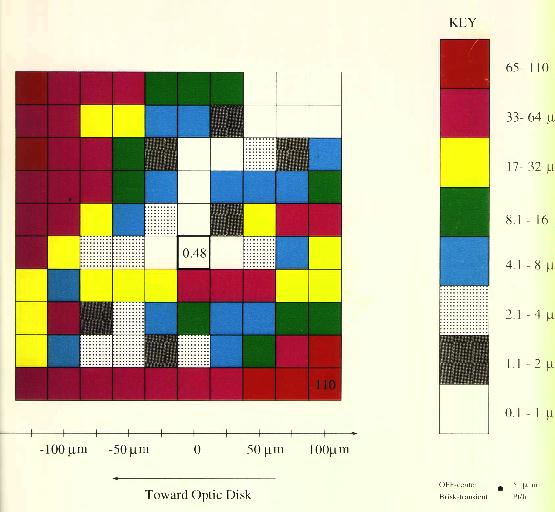
Figure 8 Cathodal Thresholds
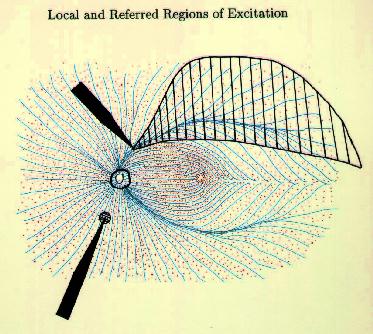
Figure 9 Local and referred regions of excitation For these reasons we have begun to investigate methods for selectively stimulating cell bodies rather than axons. Our experiments were motivated by the possibility that stimulation polarity might alter the relative thresholds of axons vs. cell bodies, as suggested in Figures 10-12 on the following page. Cathodal stimulation near the axon hillock may produce electric fields penetrating the cell body as shown in red at the left. These fields exit the cell so as to depolarize the membrane at the hillock, leading to a low stimulation threshold. But cathodal stimulation far from the axon hillock could create fields entering the cell at the hillock as shown in the center figure, leading to hyperpolarization there and elevated thresholds for cell bodies. This undesirable outcome might occur frequently, since we have no control over the location of individual electrodes on an array relative to cell body location. In such cases, the threshold might be lowered by reversing the polarity as shown below, right, causing field lines to once again exit the cell near the axon hillock. This may lower the firing threshold for ganglion cell bodies.
These experiments investigate the relation between axon and cell body thresholds using cathodal and anodal stimulation. For each recording the location was found that gave the lowest threshold using 200 microsecond anodal current pulses. The electrode was kept in that location for all subsequent stimulations of that axon or soma. To normalize the results, each soma and axon anodal threshold was divided by the average anodal soma threshold. Cathodal thresholds were similarly normalizied to the average soma cathodal threshold. Though the axon thresholds were lower than soma thresholds with cathode] stimulation, they rose to ,higher values than soma thresholds when anodal stimulation was applied, as predicted by the theory outlined above. These experiments suggest that anodal stimulation may be helpful in preferentially stimulating cell bodierather than axons. The data were obtained using 10 micron platinum-iridium monopolar electrodes (Figure 13).
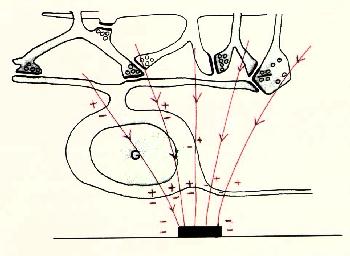
Figure 10 Cathodal Stimulation near axon hillock
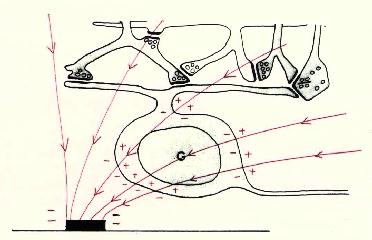
Figure 11 Cathodal Stimulation far from axon hillock
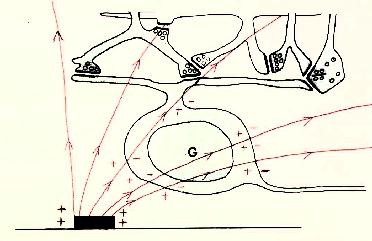
Figure 12 Anodal Stimulation far from axon hillock
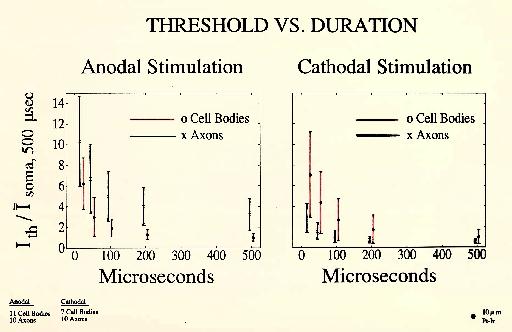
Figure 13 Anodal and Cathodal Stimulation
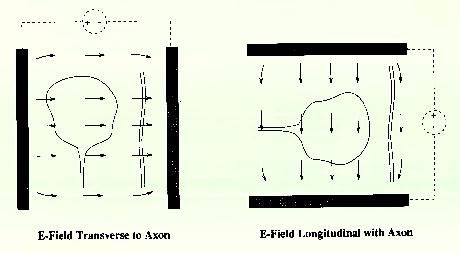
Figure 14 E-Field Transverse to Axon and E-Feild Longitudinal with Axon
In preparation for an experimental test of this idea, we have constructed a novel type of electrode (Figure 15). A 250 micron thick sheet of fused silica, coated on each side with a 5 micron gold layer, was cut into strips 0.9 mm wide and inserted into a glass tube filled with liquid epoxy. After the epoxy had hardened,the tube was cut with a wafer saw to produce a clean exposed layer, shown below.

Figure 15
Three preliminary ganglion cell stimulations were performed with this array (Figure 17). One of the electrodes was located over the receptive field center, as indicated. Thresholds were then determined for this and neighboring electrodes. A biphasic, charge-balanced current waveform helped to avoid plating out the thin gold and platinum black layers. The first and third experiments used a distant return electrode behind the sclera, while the electrodes were driven in a bipolar fashion in the second.
The third experiment gave poor localization for reasons that are under investigation, while the first and second showed good localization, with thresholds rising steeply for nearby electrodes. Electrode AffayMmsholds

Figure 16
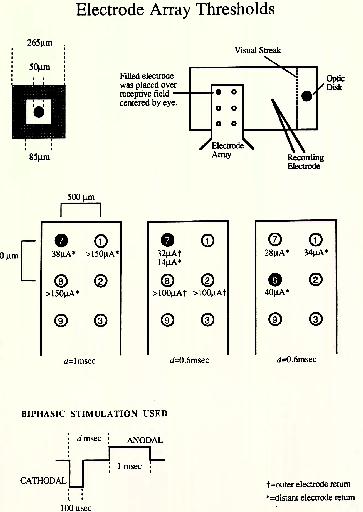
Figure 17
The thresholds rise gradually with displacements of the stimulating electrode from the soma up to 50 microns and more steeply for larger displacements. This suggests that one could hope to achieve reasonable locality for soma stimulation.
Selective stimulation of ganglion cell bodies rather than axons remains a substantial concern. We are somewhat encouraged by the improvement in relative thresholds found with anodal stimulation. We will perform experiments with non-symmetric electrodes to determine if thresholds for axons can be further raised relative to somas.